A practical procurement guide for hospital buying teams operating across multiple regions — with Umy Medical as the working example.
Most hospital procurement delays aren’t supplier problems. They’re process problems.
Consider what happened to a procurement team managing three facilities across Vietnam and the Philippines. They had a straightforward ICU equipment order — nine line items, two suppliers, nothing unusual. The hold-up wasn’t a quality issue or a lead time failure. It was a packing list discrepancy: carton weights logged at origin didn’t match the shipping manifest. The equipment sat in a bonded warehouse for 19 days while paperwork was corrected across three time zones. The clinical team had already scheduled the ward opening.
That kind of delay is almost entirely preventable. But preventing it requires more than finding a good supplier — it requires a workflow that catches documentation gaps before dispatch, not after.

This guide is written for hospital procurement officers, supply chain managers, and clinical buyers who want a structured, repeatable method to source medical equipment through a single channel.
We’ll walk through three practical controls that protect every order:
- Documentation control — what to require, what’s optional, and when to stop
- Quality control (QC) — what you can realistically verify before shipment and at receiving
- Logistics control — Incoterms, landed cost, and delivery predictability
Import, regulatory, and documentation requirements vary by destination country and facility type. Define your required documentation upfront and confirm local compliance requirements before ordering.
Why One-Stop Portals Help and Where They Fall Short
Traditional procurement forces you to manage dozens of vendor relationships simultaneously — different catalogs, different warranty language, different shipping practices. The overall system becomes fragile fast, and expensive to maintain even when individual suppliers are performing fine.
A one-stop sourcing portal reduces that fragmentation. One channel. A more consistent process. Fewer threads to track when something goes sideways.
Here’s the honest caveat, though: a portal only creates value when you add structure to it. The buyers who get strong outcomes aren’t the ones who simply “order faster.” They’re the ones who build a repeatable workflow around evidence, QC, and logistics — and require that same discipline from their sourcing partner. Speed without process just means errors arrive sooner.
Where Umy Medical Fits in This Workflow
Umy Medical functions as the coordination layer in your sourcing process: a single channel to quote, align specifications, consolidate ordering across categories, and organize commercial documentation and shipping details.
What Umy Medical supports:
- Consolidated sourcing across many product categories through one channel
- RFQ and quote handling based on your specification sheet
- Invoices and packing lists when required
- Written warranty terms and after-sales coordination based on agreed terms
- Logistics coordination and shipment details based on your order
What this is not:
- ERP integration services
- A guarantee of universal certification availability across every product and manufacturer
This isn’t a limitation unique to Umy Medical — it’s the reality of multi-region medical equipment procurement. The solution is a workflow strong enough to perform even when documentation varies.
📌 Ready to consolidate your sourcing? Send us your spec sheet and destination country. We’ll confirm product availability, document availability, and quote options — before you commit to anything. Send Us Your RFQ →
Think in “Evidence Tiers,” Not “Certificates or Nothing”
In a multi-product procurement environment, not every item comes with the same documentation package. Treating certifications as a binary pass/fail creates unnecessary bottlenecks and doesn’t reflect how professional procurement actually works.
Use a tiered evidence model instead.
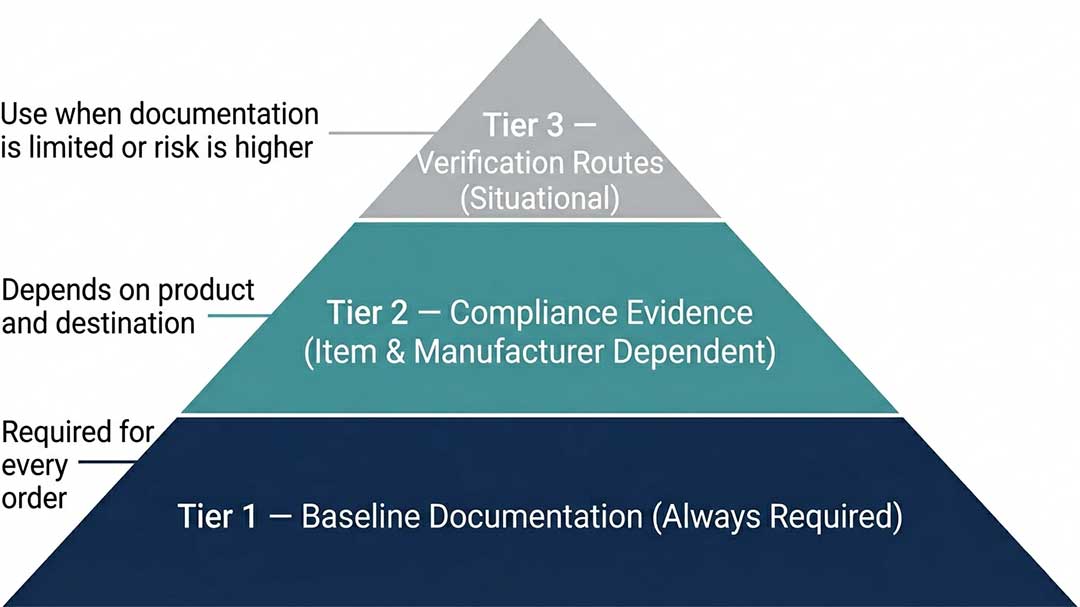
Tier 1 — The Baseline (Required for Every Order)
Tier 1 is the minimum set that makes an order auditable, receivable, and dispute-free:
| Document | Status |
|---|---|
| Quote matching your defined spec (model, version, accessories) | Required |
| Written warranty terms | Required |
| Product specs, photos, manuals (where applicable) | Required |
| Invoice and packing list | Required when applicable |
Treat Tier 1 as your standard order language with Umy Medical. Confirm all items are covered before issuing a PO.
Tier 2 — Compliance Evidence (Item- and Manufacturer-Dependent)
Depending on the product and destination, you may also request:
- ISO 13485 certificate or scope (if applicable to the manufacturer)
- CE-related documentation (where applicable)
- Device-specific test reports (when available)
Because Umy Medical coordinates products across many manufacturers, Tier 2 availability varies by item. This is why Step 5 of the workflow below includes a formal document gate — so availability is confirmed before ordering, not after.
Tier 3 — Verification Routes When Documentation Is Limited
When Tier 2 documents are unavailable — or when device risk is higher — use these verification routes:
- Sampling or evaluation orders for technical review
- Pre-shipment inspection where feasible
- Strengthened receiving acceptance checks on arrival
The rule: If a document is mandatory for your destination or internal policy and it isn’t available, you don’t work around it. You re-source — different item, different manufacturer option.
The 10-Step Procurement Workflow (No ERP Required)
A professional sourcing process doesn’t require ERP integration. It requires decision gates that hold, applied consistently from RFQ through receiving.
Step 1 — Start with the Clinical Need, Not the Catalog Page
Define the intended use and environment (ICU, OR, ward) and the device category, whether you are sourcing ultrasound equipment, surgical devices, or monitoring systems — along with minimum specifications and constraints (space, power, maintenance capacity). This prevents feature drift and stops the RFQ process from becoming guesswork.
Umy Medical tip: Send your minimum spec requirements first. Request quotes against your sheet — not against a generic model name.
Step 2 — Write a One-Page Spec Sheet
Procurement comparisons are only fair when every supplier quotes against the same document. Your spec sheet should lock: included accessories, consumables compatibility, and power requirements.
This is how you avoid “same product name, different configuration” surprises at receiving.
Step 3 — Shortlist with Acceptable Alternates
Select your preferred item and one or two alternates. This applies whether you are sourcing imaging systems, laboratory diagnostics, or ward equipment — the shortlisting logic is the same regardless of category. Decide upfront what can vary (minor features) and what cannot (core performance, safety-critical functions, compatibility).
Alternates aren’t indecision — they’re a risk-control tool when lead time or documentation availability changes.
Step 4 — Send an RFQ That Forces Clarity
A good RFQ is firm on the essentials: exact specification, quantity, destination, lead time expectation, and Incoterm preference.
With Umy Medical: an RFQ that includes destination + Incoterm preference + document requirements produces cleaner quotes and fewer late-stage surprises.
Step 5 — Document Gate: Confirm Evidence Before Placing a PO ⬅ This step saves the most time.
Stop. Before you issue a PO:
- Is your Tier 1 package confirmed?
- Which Tier 2 documents are actually available — not promised, available?
- Does device risk or a tight delivery window warrant Tier 3 verification?
If required evidence isn’t there, this is the moment to change the item or manufacturer — not after payment clears.
Practical approach with Umy Medical: include a “required documents” list in your RFQ or PO notes and request written confirmation per line item before payment or production begins.
Step 6 — Evaluate Quotes on Control, Not Only Unit Price
Unit price is one input. Also evaluate:
- Lead time realism (not just the optimistic estimate)
- Warranty terms and spare parts availability
- Packaging standard — especially for sensitive or fragile devices
- End-to-end shipping timeline
A slightly higher unit price that prevents a re-shipment or dispute is better value.
Step 7 — Place the PO with “No Surprises” Language
Your PO should lock:
- Exact model, spec, version, and included accessories
- Delivery term (Incoterm), destination, and delivery window
- Warranty terms and support expectations
- The agreed document list (including invoice and packing list when required)
The PO is your control document. Treat it like one.
Step 8 — Match QC Effort to Device Risk
Not every product needs the same QC intensity.
| Device Risk Level | Recommended QC |
|---|---|
| Lower-risk items | Photo confirmation + packaging check |
| Higher-criticality devices | Sampling, pre-shipment inspection |
| Safety-critical, high-value, or hard-to-replace | Full inspection + increased documentation |
Increase QC when documentation is limited, delivery timelines are tight, or the order is difficult to recover from if errors occur.
Step 9 — Control Shipping Documentation Before Dispatch
Customs delays are frequently caused by avoidable paperwork gaps — and this is worth saying plainly: the most common triggers we see aren’t missing certificates or regulatory failures. They’re incorrect weights, mismatched carton counts, and packing lists that don’t match the invoice. Before shipment, confirm:
- Invoice and packing list will be issued when required
- Total cartons, gross/net weight, and dimensions are accurate
- Packaging protection is adequate for fragile components
- Shipment labeling and marks are correct
Step 10 — Receive Like a Process, Not a Handshake
On arrival:
- Inspect packaging and document any damage immediately (photos)
- Verify quantities against the packing list
- For a real example of what a well-managed receiving and shipment process looks like in practice, see how we handled a patient monitors order dispatched to Azerbaijan.
- Perform basic functional checks where appropriate
- Archive all documents in one order folder: RFQ → quote → PO → shipping docs → receiving record
This step protects your facility and makes warranty and service discussions significantly easier.
📌 Want to test this workflow with a real order? Share your product category, destination, and required document list. We’ll confirm availability item-by-item before you commit. Contact Umy Medical →
Logistics Controls for Multi-Region Buyers
Incoterms — Choose Your Control Level
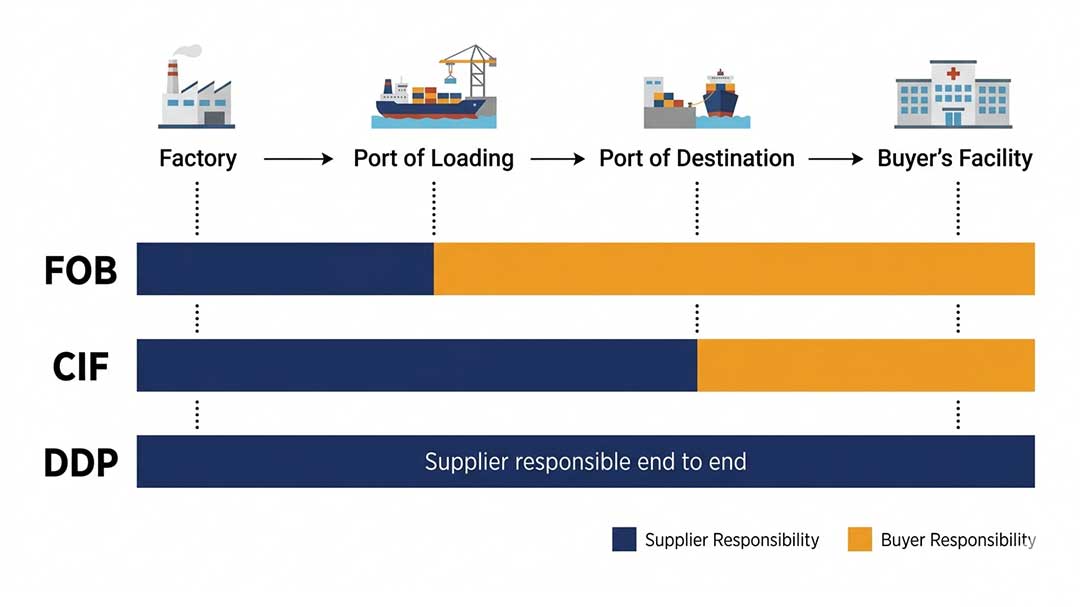
| Incoterm | Who Manages Freight | Best For |
|---|---|---|
| FOB | Buyer manages from port of loading | Teams with established freight partners |
| CIF | Supplier arranges to destination port | Simpler experience, local costs still apply |
| DDP | Delivered duties paid | Easiest experience — confirm full scope in writing |
For the full official definition of each term, refer to the Incoterms 2020 rules published by the International Chamber of Commerce.
For early orders with a new supplier, simplicity often wins — as long as the scope is explicit and confirmed in writing.
Landed Cost Surprises to Actively Prevent
A few recurring issues create unexpected spend:
- Split shipments — more brokerage fees and handling charges
- Incorrect weights or dimensions — freight billing disputes
- Missing or incorrect paperwork — customs delays and storage charges
- Rush shipping caused by late approvals — entirely avoidable premium spend
One-stop portals reduce these risks most when they reduce fragmentation: fewer suppliers, fewer partial shipments, fewer document threads to track.
When Certifications Aren’t Available
If a certification document isn’t available for a specific item, disciplined procurement follows this sequence:
- Determine whether it is mandatory for your destination, use case, or internal policy
- If mandatory → do not proceed with that item or manufacturer
- If not mandatory → reduce risk through:
- Tighter specifications and accessory lists
- Sampling or evaluation orders for higher-risk categories
- Pre-shipment inspection where feasible
- Strengthened receiving acceptance checks
- Written warranty and spare parts expectations confirmed in the PO
This approach is audit-friendly and keeps procurement in control — without pretending documentation is uniform across every product line.

RFQ Template for Umy Medical Inquiries
Use this when contacting Umy Medical. It produces faster, more accurate quotes.
Subject: RFQ – [Product Category / Model] – Delivery to [City, Country] 1) Item & Specification - Item name: - Model / version: - Key specs (must meet): - Included accessories: - Quantity: 2) Delivery - Destination: [City, Country] - Incoterm: [FOB / CIF / DDP] (or request options) - Target delivery date/window: 3) Documentation — confirm availability before PO - Tier 1 (required): quote, warranty terms, specs/manual if applicable - Commercial docs (when required): invoice, packing list - Tier 2 (if required): [list specific documents needed] - Tier 3 (optional): sampling/inspection option and cost, if available 4) After-Sales - Warranty duration: - Spare parts availability period: - Support response expectation:
The Difference Is in the Process
One-stop portals are a powerful sourcing tool for multi-region buyers — but the advantage doesn’t come from speed alone. It comes from control.
When you run a portal workflow with a clear documentation gate before the PO, QC matched to device risk, and logistics terms that prevent landed-cost surprises, orders stop being a source of operational risk and start becoming something your team can actually plan around.
How Umy Medical supports this in practice: Send your destination country and required document list — Tier 1 plus any Tier 2 requirements — and we confirm document availability item-by-item before ordering. Invoices and packing lists are provided when required.
📌 Start your sourcing inquiry today. One channel. Clear documentation. Confirmed before you commit. Send Us Your RFQ →
Import, regulatory, and documentation requirements vary by destination country and facility type. Buyers should confirm local requirements and internal policies before ordering, and define required documentation upfront.